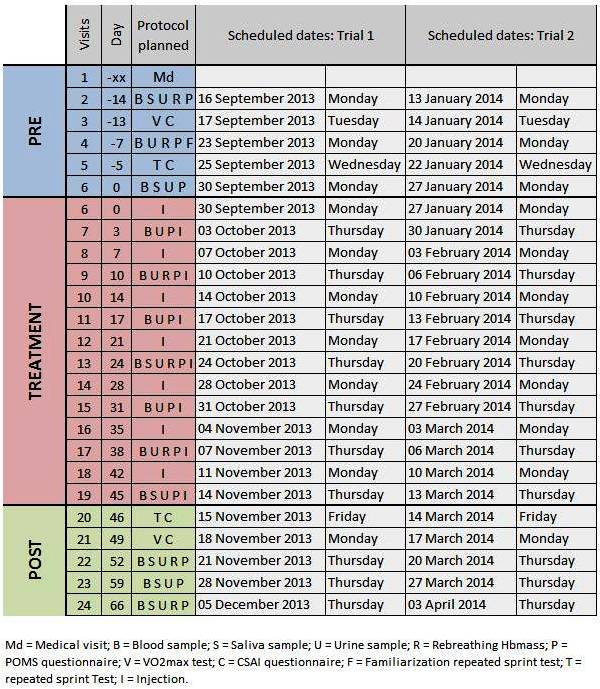
What will happen to me if I take part in this study? Initially, you will be asked to undergo a medical examination by a qualified medical practitioner to obtain information related to your general health and to establish your suitability as a subject in the study.
If selected, you will be asked to participate in a trial over a 12-week period twice (2 x 12 weeks in total). You will be randomly assigned to group 1 or 2 in a cross-over trial. In a cross-over trial, the groups each have the different treatments in turn. Group 1 will receive subcutaneous injections of recombinant human Epo twice a week for 7 weeks during the first trial, while they will receive a placebo of saline solution during the second trial; In contrast, group 2 will first receive the placebo during the first trial, while they will receive the Epo injection during the second trial. A placebo is a dummy treatment which looks like the real thing but is not. It contains no active ingredient. You will self administer all injections (just under the skin in the abdomen area) using a small needle under the supervision of trained investigators. You will not know in which treatment group you are in until the end of the entire study. The Epo doses will vary during the administration from 20 to 40 units per kilogram body weight.
As the production of red blood cells require iron and because Epo will boost the production of your red blood cells, you will also receive daily iron tablets providing approximately 65 mg of iron during the 7 weeks of Epo administration to make sure that your iron stores are adequate throughout the study. Iron will be substituted by carbonate tablets during the placebo trial.
Overall, you will be asked to come to the lab on 25 occasions in total during each trial.
On 13 occasions (once a week – time ~25 min), we would like to take a
small amount ofblood (10 mls equivalent to 2 teaspoons) from your arm
with a needle. You will be asked to provide a urine sample. Your resting
heart rate and blood pressure as well as your body weight will be
measured. You will be asked to fill up a small questionnaire to assess
how you are feeling.
On 7 occasions (once every two week – time ~45min), we would like to
take a small amount of blood (15 mls equivalent to 3 teaspoons) from
an intravenous line in your arm. In addition to the measurements
described above, you will be asked to breath a small non-toxic dose of
carbon monoxide through an equipment for measuring breathing in order
to determine your total amount of haemoglobin. Haemoglobin is part of
your red blood cell which transports oxygen from your lung to your
muscles.
On 6 occasions (once a week during the injections – time 5 min), you
will be asked to come to the lab to only receive your injection.
On 5 occasions (3 times before and twice after the injections – time
~45 min), you will be asked to perform exercise performance tests on a
laboratory bicycle, including two maximal oxygen uptake (VO2max) tests,
two repeated sprint ability tests and one familiarisation of the latter test.
Prior to each test, you will be asked to fill up a small questionnaire to
assess how you are feeling. The VO2max test consists of pedalling on
the bicycle or running on a treadmill while the load or speed is gradually
increased until exhaustion. You will wear a mask during the test which
will allow us to measure your gas exchanges. The repeated sprint ability
test consists of 10 x 10 s all-out sprints separated by a 50 s rest interval.
We would like to take a small amount of blood (15 mls equivalent to 3
teaspoons) from your arm before, during and after the repeated sprint
ability test.
You could be compensated for travel expenses and loss of earnings
according to verified documentation but you will receive no other payments.
If selected, you will be asked to participate in a trial over a 12-week period twice (2 x 12 weeks in total). You will be randomly assigned to group 1 or 2 in a cross-over trial. In a cross-over trial, the groups each have the different treatments in turn. Group 1 will receive subcutaneous injections of recombinant human Epo twice a week for 7 weeks during the first trial, while they will receive a placebo of saline solution during the second trial; In contrast, group 2 will first receive the placebo during the first trial, while they will receive the Epo injection during the second trial. A placebo is a dummy treatment which looks like the real thing but is not. It contains no active ingredient. You will self administer all injections (just under the skin in the abdomen area) using a small needle under the supervision of trained investigators. You will not know in which treatment group you are in until the end of the entire study. The Epo doses will vary during the administration from 20 to 40 units per kilogram body weight.
As the production of red blood cells require iron and because Epo will boost the production of your red blood cells, you will also receive daily iron tablets providing approximately 65 mg of iron during the 7 weeks of Epo administration to make sure that your iron stores are adequate throughout the study. Iron will be substituted by carbonate tablets during the placebo trial.
Overall, you will be asked to come to the lab on 25 occasions in total during each trial.
On 13 occasions (once a week – time ~25 min), we would like to take a
small amount ofblood (10 mls equivalent to 2 teaspoons) from your arm
with a needle. You will be asked to provide a urine sample. Your resting
heart rate and blood pressure as well as your body weight will be
measured. You will be asked to fill up a small questionnaire to assess
how you are feeling.
On 7 occasions (once every two week – time ~45min), we would like to
take a small amount of blood (15 mls equivalent to 3 teaspoons) from
an intravenous line in your arm. In addition to the measurements
described above, you will be asked to breath a small non-toxic dose of
carbon monoxide through an equipment for measuring breathing in order
to determine your total amount of haemoglobin. Haemoglobin is part of
your red blood cell which transports oxygen from your lung to your
muscles.
On 6 occasions (once a week during the injections – time 5 min), you
will be asked to come to the lab to only receive your injection.
On 5 occasions (3 times before and twice after the injections – time
~45 min), you will be asked to perform exercise performance tests on a
laboratory bicycle, including two maximal oxygen uptake (VO2max) tests,
two repeated sprint ability tests and one familiarisation of the latter test.
Prior to each test, you will be asked to fill up a small questionnaire to
assess how you are feeling. The VO2max test consists of pedalling on
the bicycle or running on a treadmill while the load or speed is gradually
increased until exhaustion. You will wear a mask during the test which
will allow us to measure your gas exchanges. The repeated sprint ability
test consists of 10 x 10 s all-out sprints separated by a 50 s rest interval.
We would like to take a small amount of blood (15 mls equivalent to 3
teaspoons) from your arm before, during and after the repeated sprint
ability test.
You could be compensated for travel expenses and loss of earnings
according to verified documentation but you will receive no other payments.
What do I have to do? You will not be able to consume any alcohol 24 hours prior to each lab visit. You will be excluded from participating in this study if you take drugs (recreational or performance enhancing drugs).
Finally and importantly, you will be ineligible to compete during the 2 x 12 week duration of the study. However, the World Anti-Doping Angency, the sponsor of the study, will not restrict subsequent participation in sporting competitions.
Jérôme Durussel | PhD Student | University of Glasgow
contact: j.durussel.1@research.gla.ac.uk | 0141 330 7377
contact: j.durussel.1@research.gla.ac.uk | 0141 330 7377